Abstract
Myelodysplastic syndromes (MDS) are a heterogeneous collection of clonal disorders of hematopoietic stem cells characterized by ineffective hematopoiesis, dysplasia, and risk of progression to secondary acute myeloid leukemia (AML). Despite the development of numerous prognostic scoring systems (MDS Comorbidity Index (MDS-CI),International Prognostic Scoring System (IPSS) and IPSS-R, updated WHO classifications, and WHO prognostic scoring system (WPSS)), little prospective data regarding the influence of these and other factors on patient outcomes is available. To better elucidate the features of early mortality in MDS we compare characteristics of patients dying within one year of MDS diagnosis with those surviving longer in our Adults in Minnesota with MDS (AIMMS) population based case-control study.
The AIMMS study is a statewide prospective population-based case-control study (R01 CA142714) conducted by the University of Minnesota (UMN), Mayo Clinic, and the Minnesota Department of Health. Adults (age 20-85 years) with a new diagnosis of MDS between April 2010 and October 2014 were recruited through the Minnesota Cancer Surveillance System. Patients completed a comprehensive questionnaire to capture retrospective epidemiologic data. All cases went through central medical review including pathology review by two independent hematopathologists, cytogenetic analyses, and oncologist medical record review with assignment of IPSS, IPSS-R, WPSS 1.2 noting past medical history, exposures, and treatment history and response. Annual clinical review of all patient cases for four years after initial enrollment is ongoing in a batched, non-real time manner.
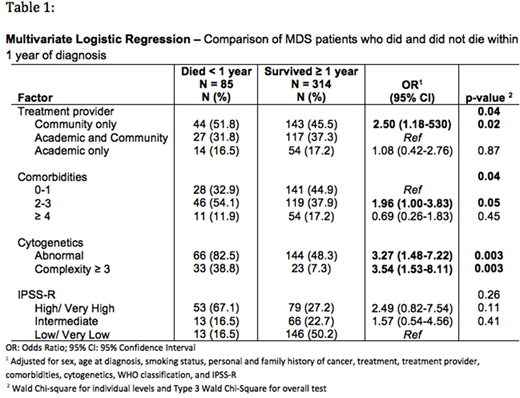
Three hundred and ninety-nine patients with a confirmed diagnosis of MDS based on central review and one year of clinical follow-up review were included in the analysis. For the entire group, we observed early mortality in those with abnormal cytogenetics, three or greater cytogenetic abnormalities, treatment at a community medical center only (versus academic), and with 2-3 concurrent medical comorbidities (Table 1). Treatment with hypomethylating agents also correlated with early mortality but when adjusted for WPSS and IPSS-R, this association was no longer significant.
To investigate varied impact of disease and patient characteristics within different groups of MDS patients, we did multivariable regression analysis within the following categories: High/Very High Risk IPSS-R; Very low/Low/Intermediate Risk IPSS-R; Treated Patients; Supportive Care Only Patients. In patients with high or very high risk IPSS-R, the presence of 3 or more cytogenetic abnormalities was the only significant predictor early mortality in a multivariable adjusted model (OR: 3.66, 95% CI: 1.57-8.53). Similarly, in those with very low, low, or intermediate IPSS-R, abnormal cytogenetics was the only significant predictor of early mortality in an adjusted model (OR: 3.71, 95% CI 1.49-9.26).
In patients receiving supportive care only, early mortality was seen in those who had diabetes (OR: 3.06, 95% CI 1.11-8.42), had complex cytogenetics (OR for ≥ 3 abnormalities: 4.94, 95% CI 1.25-19.46), and had very high or high IPSS-R (OR: 4.20, 95% CI 1.43-12.34). Likewise, complex cytogenetics remained the sole significant predictor of early mortality in treated patients (OR: 4.84, 95% CI 1.43-16.37).
Complex cytogenetics and prognostic risk category (either by WPSS or IPSS-R) have previously been associated with higher risk disease and early mortality without intervention. Our data confirm this association in a large prospectively followed MDS cohort and highlight the significance of cytogenetic abnormalities and complexity regardless of IPSS-R risk categorization or treatment.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.